Shining a Light on Optically Stimulated Luminescence
Desert Archaeology crew chief Caleb E. Ferbrache explains how electrons trapped in rock can be used to date archaeological deposits–and why, unlike the more familiar carbon dating, OSL allows dating in the absence of preserved organic material.
Most people know that archaeologists regularly use carbon-14 (also called radiocarbon) to date materials they find. While radiocarbon dating revolutionized archaeology and remains the most common dating method in the discipline, it is not the only method available. One alternative is optically stimulated luminescence.
But what is optically stimulated luminescence (OSL), other than a mouthful? Basically, it can help archaeologists figure out when things were buried by calculating the time that has passed since grains of quartz sand were last exposed to light. This is possible due to some peculiar ways that radiation interacts with quartz.

Illustration by Jerusha Shin.
The most relevant thing to know about quartz is that it is full of traps. These traps are defects in the crystal that will capture electrons. These electrons will remain trapped until exposed to an energy source, such as light or heat, which will allow the electron to escape. As the electron escapes the trap, it also releases some light. OSL gets its name because quartz is stimulated with light, and it luminesces as electrons escape.
In any case, OSL relies on the processes surrounding the capture and release of electrons in quartz to acquire a date.

Illustration by Jerusha Shin.
So, a grain of quartz destined someday to become the perfect OSL sample will begin its journey by emptying its electron traps. This typically happens through steady sunlight exposure, in a process called bleaching. Bleaching resets the OSL clock. In this example scenario, the grain is fully bleached by the sun as the wind blows it across the landscape, until it finally settles and additional wind-blown material buries it.
Once it is cut off from light, the OSL clock begins running. The sediment now surrounding the quartz will include tiny amounts of radioactive isotopes, exposing the quartz to a steady flow of ionizing radiation. The quartz captures electrons from this radiation; the radiation flow is called the dose rate. This is like the steady ticking of a clock. The quartz begins trapping electrons, and because it is cut off from light, the trapped electrons will continue building up at a fairly steady, measurable rate.
When the quartz is collected for use in OSL, it is removed from the radiation source, thereby stopping the clock. In order to date the sample, the dose rate and the equivalent dose must be identified. The equivalent dose is a calculation of the total radiation the quartz received during burial. Think of this calculation like finding a partially-filled pot underneath a dripping faucet. If you know how much water is dripping per second, and you know how much water is in the pot, you can figure out when the pot was placed under the faucet.

Caleb Ferbrache collecting OSL samples from a rockshelter in Montana. Here he is pounding a tube into the sediment for the equivalent dose sample.
The process of finding an OSL date begins with sample collection. Individual samples are collected for the equivalent dose, the dose rate, and the moisture content.
The sample for the equivalent dose must remain light-protected. This is done by hammering a metal pipe into a wall of sediment. The other two samples are collected from the area surrounding the pipe, and do not require light protection.
The sample for the dose rate is sent to a lab to identify and measure the radioactive isotopes, thereby revealing the dose rate. The moisture sample is used to identify water content. Moisture decreases radiation, so the dose rate calculation will require an appropriate correction. The dose rate is also corrected to account for contributions from cosmic rays.
Once these two samples are collected from around the pipe, the pipe is removed and sealed. It can only be opened in a dark room, where the sample is thoroughly cleaned and the quartz is isolated. At this point, the sample is ready to be placed in an OSL reader, which will perform a series of tests that should reveal the equivalent dose. The most common way to do this is a process called single aliquot regeneration (SAR).
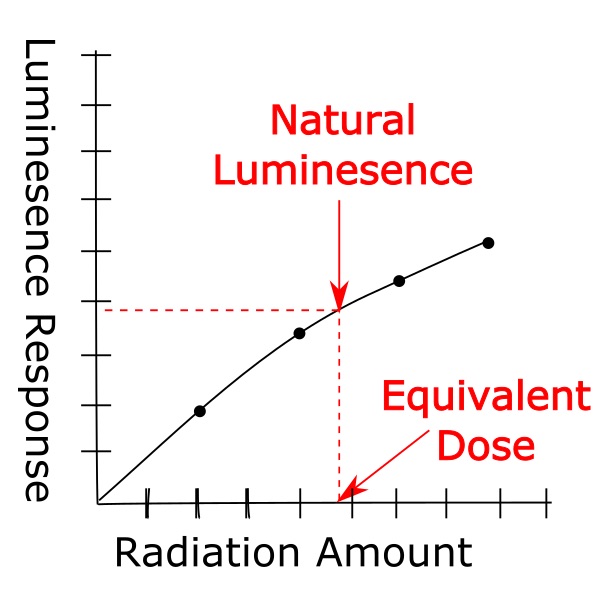
First, the reader stimulates the quartz sample with light, energizing the trapped electrons. The electrons escape and the quartz luminesces. The machine records this signal, which is casually called the natural luminescence, and it represents the total radiation the sample received during burial. The machine then performs several cycles where it exposes the sample to radiation, stimulates it with light, and measures the luminescence response. It changes the radiation amount with each cycle. It finally produces a graph that tracks the sample’s luminescence response to a range of radiation levels. The equivalent dose is at the point on the graph that matches the natural luminescence signal. Now that the equivalent dose and the dose rate are known, a date for the quartz sediment can be calculated.

The pipe for an equivalent dose sample (blue end cap visible) embedded in an exposure of canal sediments during Desert Archaeology’s excavations.
Archaeological applications for OSL are still growing, and it is becoming more popular as key advances are made. For example, Desert Archaeology’s Phoenix Office has successfully used OSL to date Hohokam canals during excavations near the Sky Harbor Airport. These dates provided a timeline that helped illuminate the canals’ construction and maintenance, as well as the natural disasters that impacted them. By acquiring dates for these events, archaeologists can think about how they fit with broader cultural and environmental observations from the same time period. Ultimately, good dates on Hohokam canals can help archaeologists understand the Hohokam people and the way they responded to the unique challenges presented by life along the Salt River.
Without good dates, archaeological questions are hard to answer.
Resources
Ferbrache, Caleb (2019) Finding the Time: Age-Depth Models in Rockshelters and their Paleoenvironmental Implications
Munyikwa, Ken (2014) Luminescence Chronology
Nelson, Michelle et al. (2015) User Guide for Luminescence Sampling in Archaeological and Geological Contexts